AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Molecular orbital theory diagram1/14/2024  Li 2 + and Li 2 – ions have the same bond order ( 0.5).īut Li 2 – has more electrons in higher energy antibonding molecular orbital as compared to Li 2 +. Li 2 + is more stable as compared to Li 2 – : Thus lithium molecule (Li2) is more stable. (d) The bonding molecular orbitals have more of the. Therefore, Li2 has stronger single covalent bonding. (c) Their molecular orbital diagrams are more symmetrical than those of homonuclear diatomic molecules. Since the bond order of Li 2 is higher than Li 2 + and Li 2. The antibonding molecular orbital σ*2 s contains 1 electron.īond order = Number of electrons in BMO – Number ofīond order = 0.5 Arrange the following species in increasing order of stability: Li 2, Li2 +, Li 2 – Chemistry Class important question MO diagram Stability order of Li2,Li2-,Li2+ In the case of Li 2 -, the 2 electrons are present in σ 2s orbitals. In the case of Li 2 +, the 1 electrons are present in σ 2s orbitals.īond order = Number of electrons in BMO – Number of electrons in ABMO / 2 As can be seen from the energy diagram - four of the molecular. How to draw the molecular orbital diagram of Li 2, Li2 +, Li2 – ? MO diagram MO diagram of Li2 ,Li2+ ,Li2- The ethane molecule has fourteen valence electrons occupying seven bonding molecular orbitals. The antibonding molecular orbital σ*2 s is empty.īond order = Number of electrons in BMO – Number of electrons in ABMO /2īond order 1 shows that there is a single covalent bond between the lithium atoms in the Li 2 molecule. The 2 electrons are present in σ 2s orbitals. How do draw the Molecular Orbital Diagram of Lithium molecule (Li 2)?Īccording to the molecular orbital theory, the molecular orbital diagram of the lithium molecule is shown below: MO diagram Li2 bond order The electronic configuration of the Li atom is 1s 2 2s 1. The distribution of electrons in lithium electronic shells is as follows: The atomic number of Lithium (Li ) is 3 and the mass number is 6.941. Lithium is represented by the symbol “Li”. 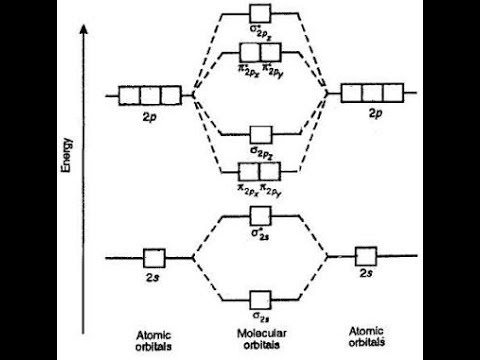
Lithium belongs to group 1 ( alkali metals), period 2 s-block element of the periodic table. How to draw Molecular Orbital Diagram of Li 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
